
The entropy of a substance can be obtained by measuring the heat required to raise the. D)ΔG<0 because although entropy decreases because the number of gaseous product particles is less than the number of gaseous reactant particles, energy is released as the bond between the H and Cl atoms forms. At absolute zero (0 K), the entropy of a pure, perfect crystal is zero. 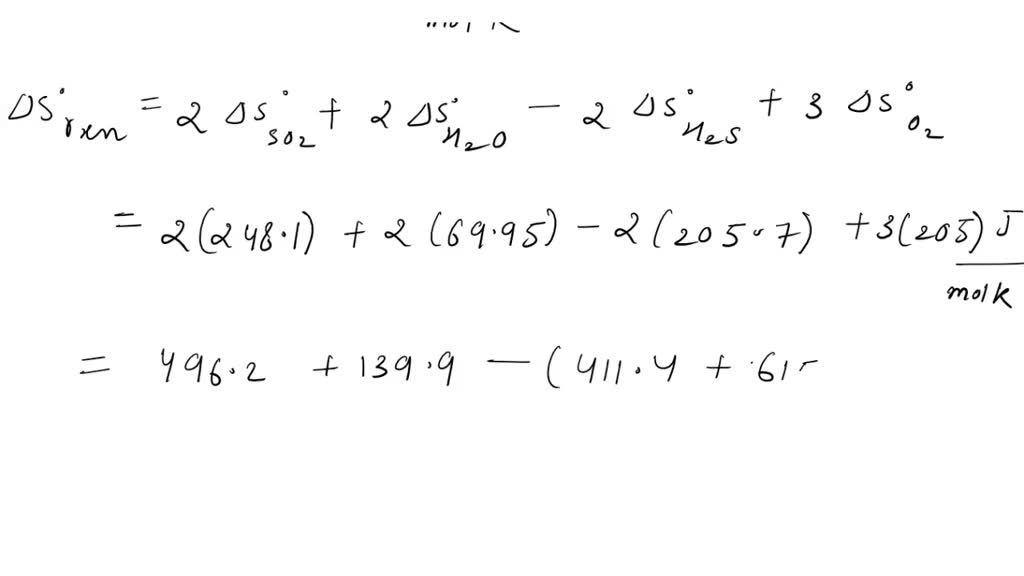
Using standard absolute entropies at 298 K, calculate the entropy change for the. C) ΔG<0 because although energy is absorbed as the bond between the H and Cl atoms forms, entropy increases because the number of gaseous product particles is less than the number of gaseous reactant particles. We show, for both organic liquids and solids, that entropies are reliably linearly correlated with Volume per molecule, V-m (nm(3) per molecule) (or molar. Question: Consider the reaction:2H2O (l) 2H2 (g) + O2 (g)Using standard absolute entropies at 298 K, calculate the entropy change for the system when 2.19 moles of H2O (l) react at standard conditions.Substance S (J/k times mol)H2 130.7O2 205.1H2O 69.9 J/K. B) ΔG>0because energy is absorbed as the bond between the H and Cl atoms forms, and entropy decreases because the number of gaseous product particles is less than the number of gaseous reactant particles. What are the values of AH, AG, and of the absolute entropy, S, for H at 25C. Absolute Entropies Energy values, as you know, are all relative, and must be defined on a scale that is completely arbitrary there is no such thing as the absolute energy of a substance, so we can arbitrarily define the enthalpy or internal energy of an element in its most stable form at 298K and 1 atm pressure as zero. Which of the following best explains why the reaction is thermodynamically favored? A) ΔG>0 because energy is released as the bond between the H and Cl atoms forms, and entropy increases because the number of gaseous product particles is less than the number of gaseous reactant particles. You take the entropy of water vapor at 100 C and 1 atm., and then use the ideal gas heat capacity at constant pressure to then get the entropy at the hypothetical state of 298 and 1 atm using dSCpdT/T. H(g)+Cl(g)→HCl(g) The formation of HCl(g) from its atoms is represented by the equation above. 298 K and 1 atm is a hypothetical state for pure water vapor, based on treating the water vapor as an ideal gas. 
Because ΔH is large and negative, the reaction will be not be thermodynamically favorable at any temperature. D)There are more particles (including particles in the gas state) in the reactants than in the product, thus ΔSrxn>0. Absolute entropy is a fundamental property of matter at a specified set of. Because ΔH is large and negative, the reaction will be thermodynamically favorable at all temperatures. at 298 K, AG-176.9 kJ - 298 K(-56.77 x 10-3 kJ/K) -159.98-160.0 kJ. This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Which of the following correctly explains whether or not the reaction is thermodynamically favorable? A) There are more particles (including particles in the gas state) in the reactants than in the product, thus ΔSrxn0. Using standard absolute entropies at 298K, calculate the entropy change for the system when 1.80 moles of NO(g) react at standard conditions. ΔS° is positive, as expected for a combustion reaction in which one large hydrocarbon molecule is converted to many molecules of gaseous products.4Fe(s)+3O2(g)⇄2Fe2O3(s)ΔH=−1,650kJ/molrxn The oxidation of Fe(s) is represented by the chemical equation above.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
