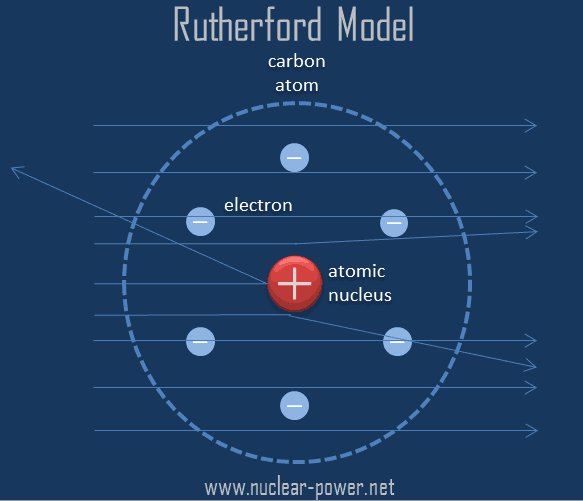
NOTE: You may want have students answer question 1 on The Smallest Matter Worksheet first. You can also search online and show students an image, simulation or video of Rutherford’s gold foil experiment when conducting this activity. Have students work in pairs or small groups to simulate Rutherford’s gold foil experiment and to answer the questions on The Smallest Matter Worksheet.They formed their model after Rutherford conducted a gold foil experiment (described in the The Radioactive Atom: Teacher Background Information). Explain that Ernest Rutherford and Niels Bohr were among those early scientists who wanted to know more about atoms. Provide students with a copy of The Smallest Matter Worksheet.For example, scientists are exploring how to use nuclear fusion - the joining of lighter nuclei to create a larger one - to generate power. Beginning with early science scholars throughout history and into this century, scientists strive to learn more about the atom and how to control it. Whether we know everything about atoms.Early scientists started forming theories about, and conducting experiments to confirm, the existence and structure of atoms. Ancient Greeks were the first to believe that all matter in the universe must be made of tiny building blocks - or atoms. How we know about the existence of atoms and their structure.Ask students to explain or hypothesize:.Start with a vocabulary activity if students are not familiar with radiation and the terms used in this activity, or provide students with the terms and definitions.Sources may include TeacherTube or other allowed Internet sourcesĤ5-60 minutes, not including optional activities or extensions. 
Online video of the gold foil experiment (optional).Marbles, ping pong balls or other small balls to represent alpha particles being shot through the gold foil.Small solid objects to represent the protons within the nucleus of gold atoms (e.g., small gravels or rocks that have a flat bottom to keep them in place).Solid object to represent the detecting screen (e.g., thin metal sheet, cardboard or books).Objects to simulate the gold foil experiment (enough for a class or group demonstrations).The Smallest Matter Worksheet (one per student, pair or group) and The Smallest Matter: Teacher Answer Key.The Radioactive Atom: Teacher Background Information.Structure and Properties of MatterĮach italicized document title can be found at the bottom of this page, and is available for printing and distribution. The concepts in this activity can be used to support the following science standard: NOTE: This activity serves as an introduction to atomic structure and does not address radiation or radioactive elements. Explain how the Rutherford-Bohr Theory of Atomic Structure helps provide us with a basic understanding of atomic structure.Simulate Ernest Rutherford’s Gold Foil Experiment.Printable Worksheets and Classroom Aids.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
